UK authorizes Pfizer vaccine for emergency use
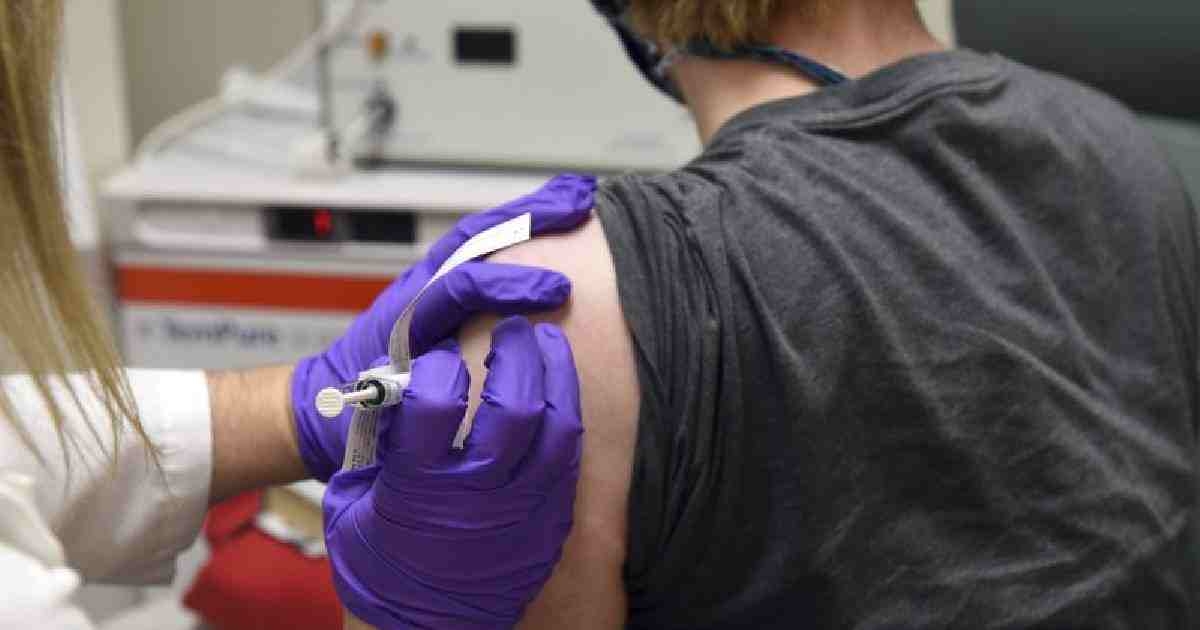
Pfizer and BioNTech say they've won permission Wednesday for emergency usage of their COVID-19 vaccine in Britain, the world’s first coronavirus shot that’s backed by rigorous science -- and a significant step toward eventually closing the pandemic.
The move makes Britain among the first countries to begin vaccinating its population since it tries to curb Europe’s deadliest COVID-19 outbreak.
Other countries aren’t much behind: The U.S. and europe also are vetting the Pfizer shot plus a similar vaccine made by competitor Moderna Inc.
Pfizer said it could immediately begin shipping limited products to the U.K. -- and provides been gearing up for even wider distribution if presented a similar nod by the U.S. Food and Drug Administration, a decision expected as soon as next week.
But doses everywhere are scarce, and initial items will come to be rationed until even more is made in the first almost a year of next year.
Pfizer CEO Albert Bourla called the U.K. decision “a historic minute.”
“We are concentrating on moving with the same level of urgency to safely source a high-quality vaccine all over the world,” Bourla said in a declaration.
While the U.K. possesses ordered more than enough Pfizer vaccine for 20 million people, it’s not clear just how many will arrive by year’s end and increasing the distribution challenges is definitely that it should be kept at ultra-cold temperatures.
Two doses three weeks apart are required for protection. Primary in brand, the U.K. government says, are frontline healthcare workers and nursing residence residents, accompanied by older adults.
British regulators are also considering another shot made by AstraZeneca and Oxford University. But Prime Minister Boris Johnson possesses warned “we should first navigate a hard winter” of restrictions to try to curb the virus until there’s enough vaccine to bypass.
Every country has several rules for determining when an experimental vaccine is effective and safe plenty of to use. Intense political pressure to become the first to roll out a rigorously scientifically analyzed shot colored the race in the U.S. and Britain, even while researchers pledged to cut no corners. On the other hand, China and Russia possess offered different vaccinations to their citizens before late-stage testing.
The shots created by U.S.-established Pfizer and its own German partner BioNTech were tried in thousands of people. Even though that study isn’t full, early results advise the vaccine is 95% able to preventing mild to severe COVID-19 disease. The firms informed regulators that of the initial 170 infections detected in study volunteers, just eight were among people who’d received you see, the vaccine and the rest got gotten a dummy shot.
“That is an extraordinarily strong protection,” Dr. Ugur Sahin, BioNTech’s CEO, recently advised The Associated Press.
The companies also reported no serious side effects, although vaccine recipients may experience non permanent pain and flu-like reactions soon after injections.
But experts caution a vaccine cleared for emergency make use of continues to be experimental and the ultimate testing should be completed. Even now to be determined is usually if the Pfizer-BioNTech shots drive back persons spreading the coronavirus without displaying symptoms. Another problem is how prolonged protection lasts.
The vaccine also offers been tested in only a tiny number of children, none younger than 12, and there’s no information on its effects in women that are pregnant.
